The pharmaceutical manufacturing execution system (MES) market is on a steep growth trajectory - projected to reach USD 4.67 billion by 2030, up from USD 2.37 billion in 2025, at a CAGR of 14.3% - and M&A activity is reshaping the competitive landscape well ahead of that inflection point. The latest signal: Title21 Health Solutions announced on January 6, 2026, the acquisition of MyCellHub and its MES platform, expanding its ability to connect the advanced therapies ecosystem through digital solutions.[1]
The deal positions Title21 as a vertically integrated software provider for the cell and gene therapy (CGT) sector - a market where data integrity failures carry direct patient risk and regulatory deficiencies can halt manufacturing entirely. For operations and IT leaders at hospitals, contract development and manufacturing organizations (CDMOs), and advanced therapy developers, the transaction raises substantive questions about platform interoperability, compliance continuity, and vendor consolidation risk.
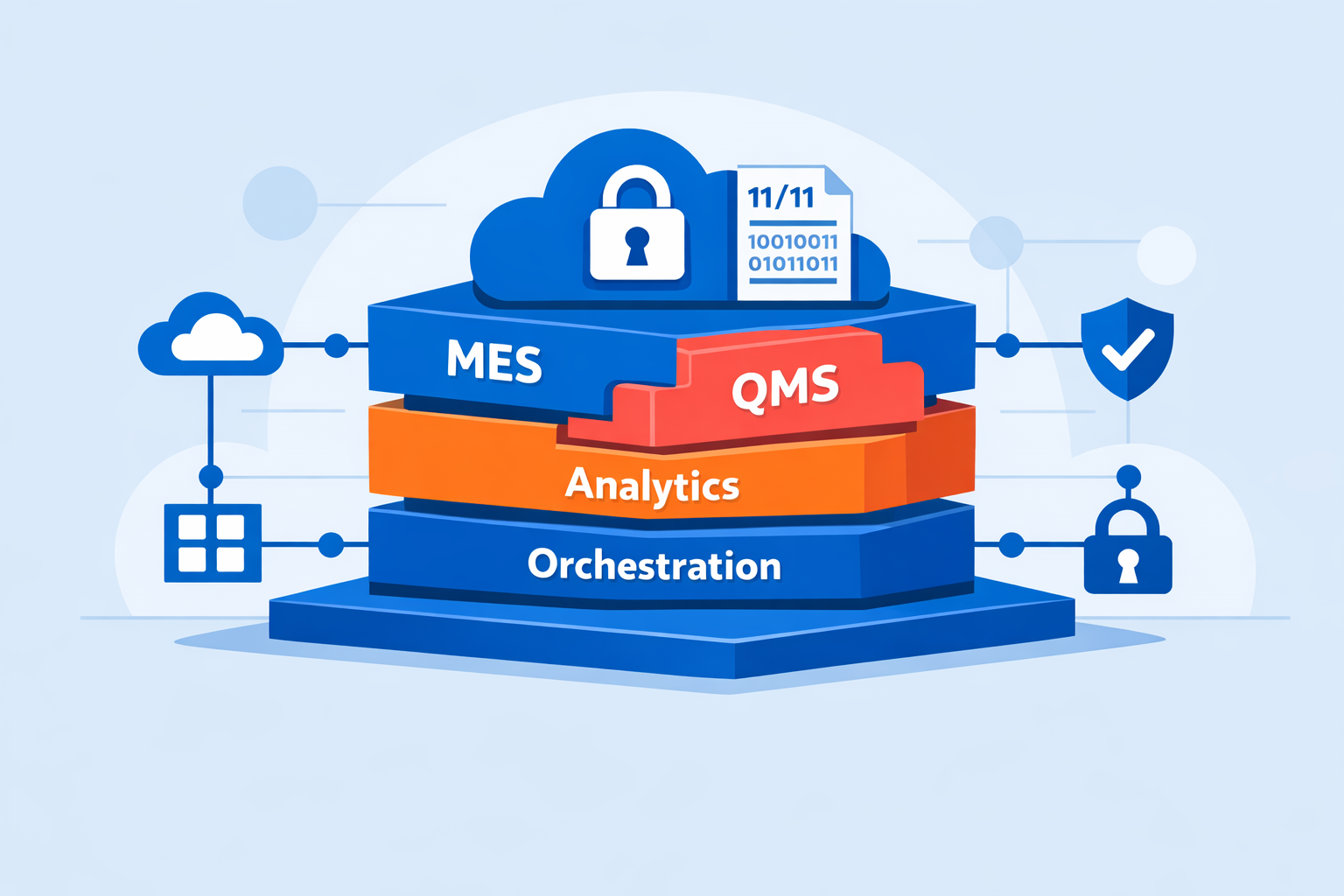
The Deal: From Point Solution to Single-Source Platform
Headquartered in Hasselt, Belgium, and led by CEO Jacques Parlongue, MyCellHub provided cloud-based MES technology for regulated life science and advanced therapy manufacturing. The platform enabled organizations to transition from paper-based processes to secure, 21 CFR Part 11-compliant digital batch records and production workflows, ensuring data integrity, process control, and regulatory compliance.
Financial terms of the transaction were not disclosed. Title21 is an ArchiMed portfolio company focused on delivering integrated technology to support healthcare providers and the health sciences.
With a MES platform now in its portfolio, Title21 is positioned to become a single-source provider for standard of care (hematopoietic cell transplant), advanced biotherapy manufacturing, integrated analytics, and quality management systems. The geographic dimension is equally significant: the acquisition extends Title21's coverage in North America and Australia to include Europe, with the Belgium-based MyCellHub team joining Title21's workforce.
From a market consolidation standpoint, the deal reflects a broader pattern. Healthcare IT dealmaking rose in 2025, reaching four-year highs in volume and value as providers, payers, and investors sought digital tools to manage costs, improve efficiency, and augment hard-to-find staff. Buyers focused on advanced pharma manufacturing and integrated platforms, with companies building digital partner networks to manage complexity and support future growth.
Compliance Architecture: What Changes for Existing MyCellHub Customers
The central compliance question for current MyCellHub users is whether post-acquisition platform integration will trigger revalidation obligations under FDA Computer Software Assurance (CSA) guidance and 21 CFR Part 11.
MES systems enforce SOPs, capture detailed production data, maintain electronic records, and facilitate audits and inspections - helping organizations adhere to regulations such as 21 CFR Part 11 and GMP guidelines. MyCellHub's platform was purpose-built to these requirements, enabling GMP teams to move from paper-based processes to secure, compliant digital batch records and production workflows while improving operational efficiency, enhancing quality oversight, and scaling complex manufacturing processes to meet evolving regulatory and business needs.
Post-acquisition product roadmap changes - such as platform migration, API refactoring, or merging of the MES with Title21's QMS or orchestration layers - may constitute "significant changes" under CSA guidance, potentially requiring updated impact assessments, User Acceptance Testing (UAT) documentation updates, and traceability matrix reviews.
Key consideration for validated system owners: Any modification to a 21 CFR Part 11-compliant system that alters electronic record creation, audit trail behavior, or electronic signature workflows typically triggers a validation impact assessment. Customers should request a formal Software Change Control Notification from Title21 before any major platform updates are applied.
The regulatory environment is also tightening independently of this transaction. European GMP regulators are revising Annex 11 in 2025, with a draft that expands emphasis on lifecycle traceability, stricter audit trail requirements (audit trails must be always on and locked), and mandatory multi-factor authentication. In pharmaceutical manufacturing, compliance with 21 CFR Part 11 is critical for ensuring the integrity of electronic records and signatures within MES. Comprehensive frameworks must now align with 21 CFR Parts 11, 210, 211, ICH Q7, and EudraLex Volume 4 Annex 11 - encompassing system design, user access controls, audit trails, and data lifecycle management.
Traceability and Lot-Level Lineage in Cell Therapy: The "Arm-to-Arm" Imperative
Cell and gene therapy manufacturing operates under a uniquely demanding traceability model. Unlike conventional pharmaceutical batch production, autologous and allogeneic therapies require unbroken chain-of-identity (CoI) documentation from the moment of donor/patient collection through cleanroom manufacturing, quality release, and clinical infusion - the so-called "arm-to-arm" workflow.
MyCellHub's MES enables Title21 to close the gap between the cleanroom and the rest of the therapy lifecycle, from collection through infusion. This is not a trivial claim: any data discontinuity in the cleanroom-to-infusion chain can trigger a chain-of-identity deviation, potentially disqualifying a batch or delaying patient treatment.
The MES technology is specifically designed to meet the stringent requirements of the healthcare, biotech, and cell therapy sectors by delivering real-time production monitoring, digital batch record management, and embedded quality management within manufacturing operations.
For contract manufacturers and CDMOs, lot-level lineage requirements extend further - encompassing raw material genealogy, starting material release, in-process testing, and finished product serialization. MES systems improve traceability by providing a detailed digital history of the production process, including raw materials, components, and manufacturing steps, which is essential for quality control, recalls, and regulatory audits.
The integrated Title21 + MyCellHub stack is positioned to address these requirements across the full therapy lifecycle. However, the depth of actual data integration - versus a loosely coupled portfolio of separate products - will determine whether that promise is realized.
Interoperability Standards and Product Roadmap Considerations
The acquisition raises important interoperability questions for organizations that have deployed MyCellHub alongside third-party EHR/HIS platforms, laboratory information management systems (LIMS), or supply chain management tools.
| Capability | Pre-Acquisition (Standalone MyCellHub) | Post-Acquisition (Title21 + MyCellHub) |
|---|---|---|
| Scope | MES - GMP batch records, process control | MES + QMS + orchestration + analytics |
| Traceability coverage | Cleanroom / manufacturing execution layer | Arm-to-arm: collection -> manufacturing -> QC -> infusion |
| Geographic reach | Europe-focused | North America, Australia, and Europe |
| Compliance stack | 21 CFR Part 11, GMP Annex 11 | Part 11, Annex 11, ATMP-specific QMS frameworks |
| EHR/HIS connectivity | MES-centric integrations | Expanded via Title21's orchestration layer |
| Single-source potential | No - separate QMS and analytics required | Yes - QMS, MES, orchestration, analytics unified |
Key roadmap questions for procurement and IT/OT teams:
- Will existing MyCellHub API endpoints and integration connectors be deprecated during platform unification?
- What is the timeline for any required data migration between MyCellHub's existing cloud infrastructure and Title21's hosting environment?
- How will Title21 handle versioning and backward compatibility of electronic batch record schemas across the merged stack?
- Will the combined platform support FHIR-based interoperability with hospital EHR systems for direct therapy order and chain-of-identity data exchange?
Industry analysts expect demand for healthcare IT to remain strong as organizations across the spectrum prioritize interoperability, automation, and value-based care capabilities to navigate demographic shifts and margin pressures. For regulated manufacturing customers specifically, interoperability is not a differentiator - it is a baseline operational requirement.
Vendor Ecosystem Risk: OT/IT Security and Accountability in a Consolidated Stack
Any MES vendor consolidation event introduces a transient period of elevated OT/IT security and governance risk. Key risk vectors for organizations currently using MyCellHub or evaluating the combined Title21 platform include:
1. Change in Security Posture Post-acquisition cloud infrastructure migrations, authentication system mergers, and network topology changes can introduce temporary access control gaps. Organizations should conduct a fresh penetration testing cycle and update Supplier Security Assessment questionnaires to reflect the new vendor entity.
2. Data Governance Continuity Legal diligence in healthcare IT deals increasingly centers on data ownership, interoperability, cybersecurity, and regulatory compliance under HIPAA and emerging AI governance frameworks. Existing MyCellHub customers should review data processing agreements to confirm that data residency, retention, and access provisions remain enforceable under the Title21 organizational umbrella - particularly given MyCellHub's Belgian domicile and applicable GDPR obligations.
3. Support and SLA Continuity Acquisition periods frequently see attrition of key engineering and support personnel. Manufacturing IT and quality teams should establish named support escalation paths in the new organization and re-confirm SLA terms for critical incident response, particularly for systems that gate batch release or patient scheduling.
4. Audit Readiness During Transition Stringent regulatory frameworks such as US FDA 21 CFR Part 11, EU GMP Annex 11, PIC/S guidelines, and global data-integrity mandates are significantly accelerating MES adoption in the pharmaceutical sector - and manufacturers must ensure complete traceability, audit readiness, electronic signatures, and error-free batch documentation to comply. Any system transition that creates gaps in audit trail continuity can generate findings during regulatory inspections.
Strategic Takeaways for Healthcare Manufacturing Leaders
For current MyCellHub customers:
- Request a Software Change Notification protocol and formal roadmap documentation from Title21 covering the next 12-18 months
- Initiate a validation impact assessment for any planned platform updates within 90 days of acquisition close
- Review data processing agreements and GDPR/HIPAA data residency clauses for updated compliance under the new parent company
For organizations evaluating MES for cell and gene therapy:
- The Title21 + MyCellHub stack is now among the most vertically integrated offerings in the ATMP manufacturing software space - but procurement teams should request detailed API documentation and integration architecture diagrams before assuming seamless interoperability
- Evaluate whether a single-vendor strategy aligns with the organization's risk tolerance for vendor concentration, particularly in a sector where regulatory inspections can reference system deficiencies directly
For CDMOs and contract manufacturers:
- Assess whether the combined platform's lot-level lineage and serialization capabilities meet multi-product, multi-client traceability requirements - including the ability to maintain strict data segregation between client programs
The broader M&A environment in life sciences IT suggests this will not be an isolated deal. In 2025, the life sciences deal landscape was shaped by strategic transactions targeting proven assets in oncology, diagnostics, and next-generation manufacturing, with stakeholders prioritizing clinical value, technological sophistication, and the ability to navigate regulatory and market uncertainty. MES platforms at the intersection of manufacturing operations and patient safety will continue to attract consolidation interest - making vendor accountability frameworks and interoperability standards an increasingly critical component of technology strategy for regulated manufacturers.
For further context on data governance models driving MES scalability across industrial environments, see the analysis of federated governance and vendor consolidation in enterprise MES deployments.
Frequently Asked Questions
Does the Title21 acquisition of MyCellHub require existing customers to revalidate their MES? Not automatically. However, any post-acquisition changes to software functionality, electronic record structures, or system architecture may trigger a validation impact assessment under FDA Computer Software Assurance (CSA) guidance. Customers should maintain proactive change control dialogue with the vendor.
What regulatory frameworks govern the MyCellHub MES platform? MyCellHub was designed for compliance with 21 CFR Part 11 (FDA electronic records and electronic signatures), GMP guidelines, and EU GMP Annex 11. The 2025 draft revision of Annex 11 adds requirements for always-on audit trails, mandatory multi-factor authentication, and lifecycle traceability that customers should assess against their current configurations.
What is "arm-to-arm" traceability in cell therapy manufacturing? In autologous cell and gene therapy, every unit of therapy is derived from a specific patient or donor. "Arm-to-arm" traceability refers to the unbroken chain-of-identity documentation linking donor collection through GMP manufacturing, quality control, and clinical infusion - ensuring the correct therapy is administered to the correct patient. Any data discontinuity in this chain represents both a regulatory and a patient safety risk.
How does MES vendor consolidation affect OT/IT cybersecurity posture? Acquisitions can introduce temporary vulnerabilities through infrastructure migrations, authentication system changes, and support personnel transitions. Affected organizations should update supplier security assessments, verify data residency and access controls under the new vendor entity, and re-confirm incident response SLA terms.
Is the pharmaceutical MES market continuing to grow despite consolidation? Yes. The pharmaceutical MES market is projected to reach USD 4.67 billion by 2030, up from USD 2.37 billion in 2025, at a CAGR of 14.3%. Stringent regulatory compliance, rising biologics and vaccine production, and demand for electronic batch records are cited as key drivers of pharmaceutical MES market growth. Consolidation is reshaping the competitive landscape but not constraining overall adoption.